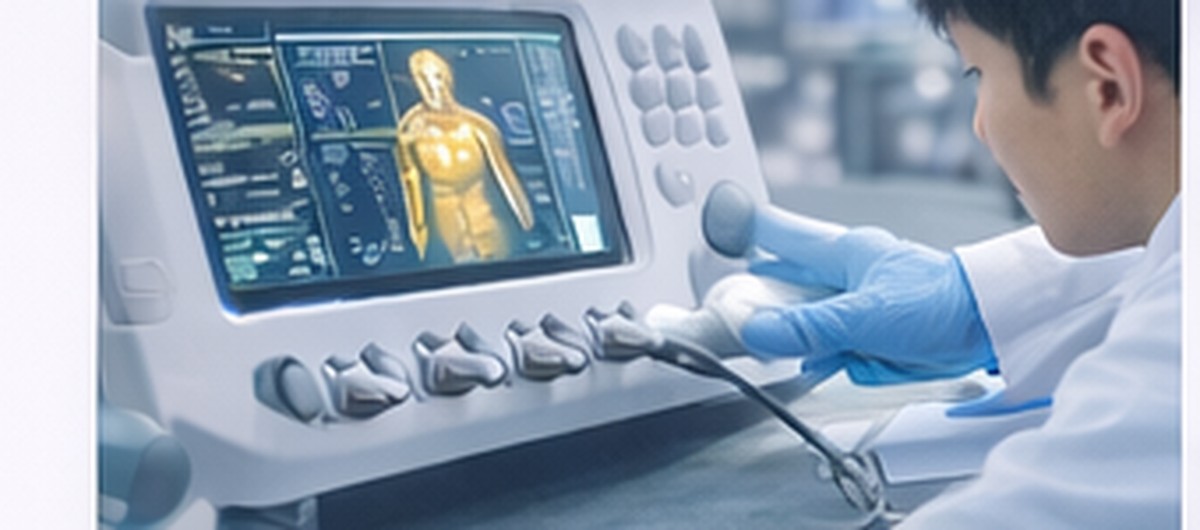
PEMF Device
We support system architecture design with coil/power/drive-waveform design and safety considerations.
We support development of medical electronic devices based on PEMF, electrical stimulation, sensors, and control technology—from design to prototyping and verification readiness, with clear deliverables and documentation.


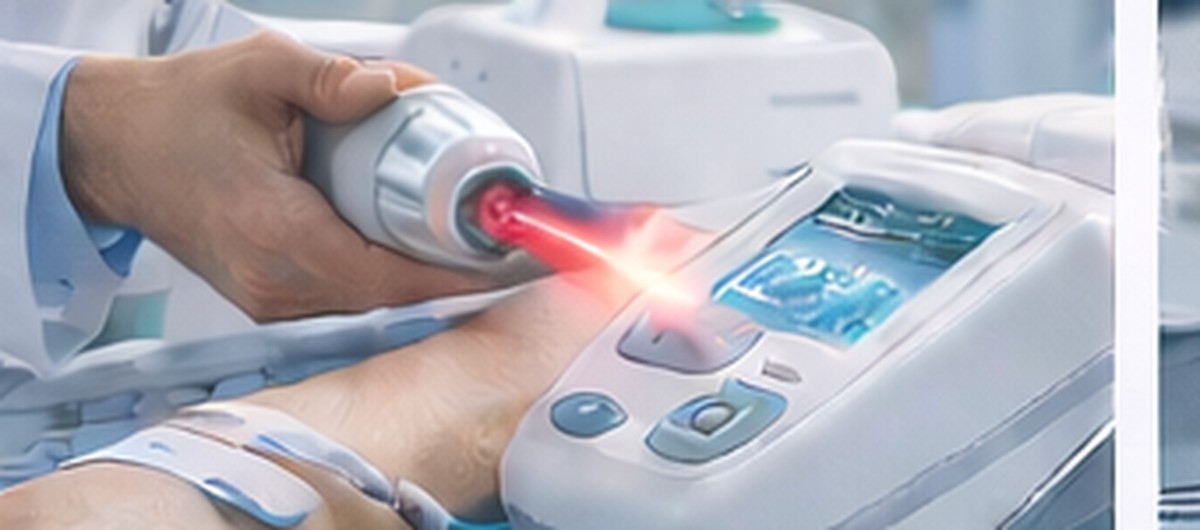
Equipment · Measurement · Electronics Design

We support system architecture design with coil/power/drive-waveform design and safety considerations.
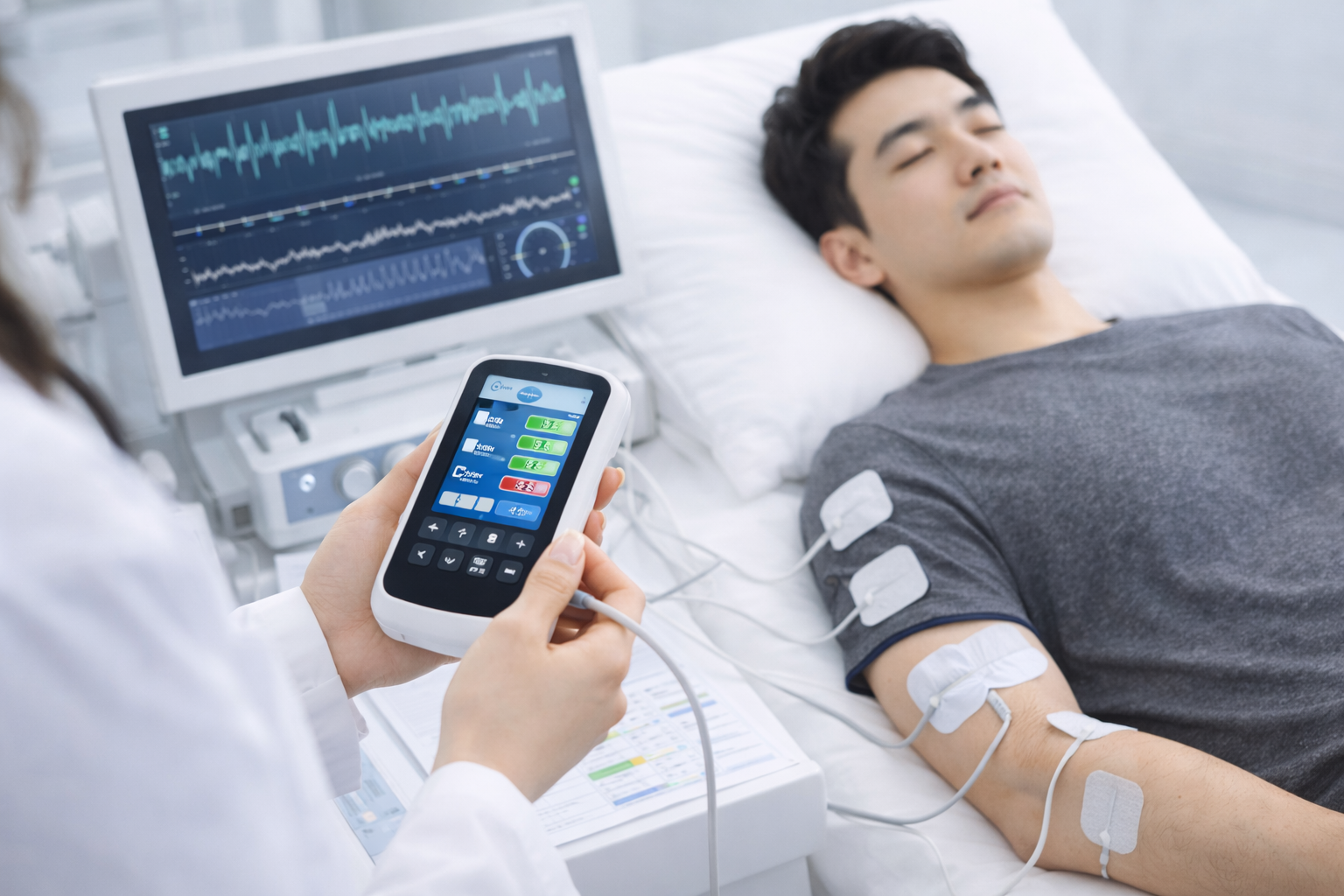
We deliver development outputs including stimulation parameter definition, sensor integration, waveform control, and logging.

We organize measurement points, test scenarios, data collection, and report templates.

We support integrated design across PCB/power/sensor interfaces, MCU firmware, and UI/control logic.

We align a productization direction considering heat/noise/durability, assemblability, and manufacturability.

We structure development documentation—risk/requirements/verification docs—based on project scope.
From coil and drive waveforms to safety/EMC—organized by development stage.
We support coil design to meet target output, considering inductance/resistance/heating and magnetic-flux distribution.
We design parameter definitions (frequency/duty/intensity limits, etc.) and firmware control/logging architecture.
We incorporate design directions that account for overcurrent/overtemperature protection and EMI/EMC issues from the start.
Depending on scope, we support the documents and verification readiness required in development.